|
12/28/2023 0 Comments Chemdoodle help formal charge 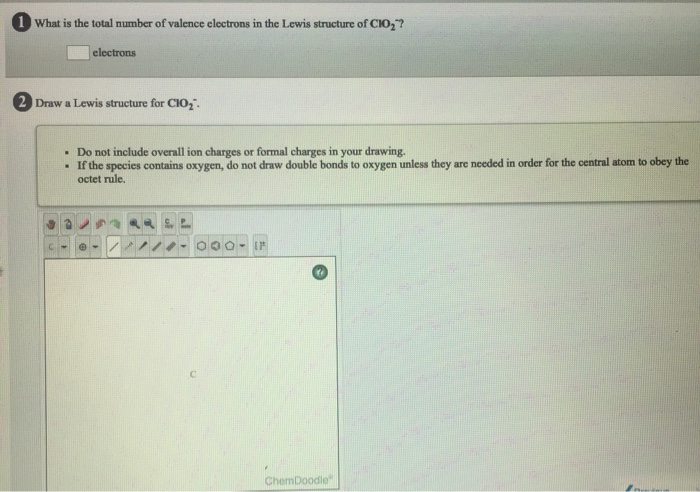
Use the dropdown menu in the bottom right corner to add a sketcher for the second molecule, and separate the two sketchers with a+ sign. To complete this exercise, try drawing both of the representations of the sulfate ion shown above. The center-left number defines the multiple count. The bottom-right number defines the repeat count. The top-right number defines the charge amount. Drawing Isomers of Organic Molecules: Practice Problems please help Name the. To show the formal charge on an atom in ChemDoodle, use the + or -tools that have been described previously, lon charge: The overall ion charge is a property of. Pressing the up and down arrows will increment and decrement the values at these positions. Lewis Structure (use A as the central atom and X as the bonded atoms). To show the formal charge on an atom in ChemDoodle, use the + or - tools that have. For example, in the drawing of the sulfate ion (SO42-) shown on the left, each oxygen atoms is assigned a formal charge of minus one and the sulfur atom is assigned a formal charge of plus two. 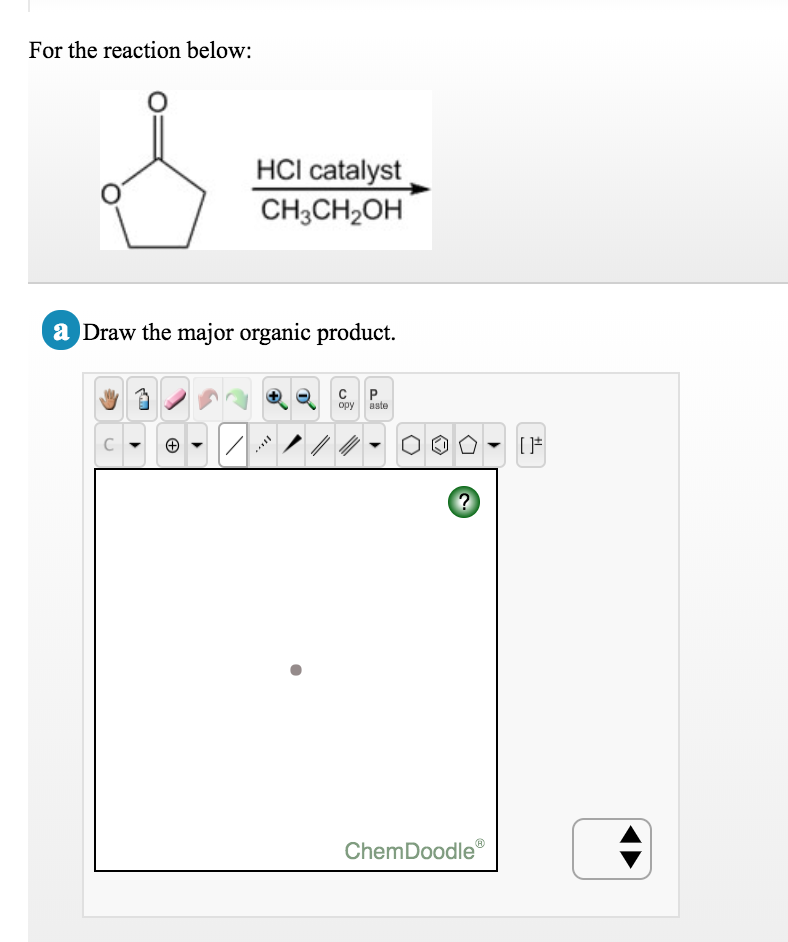
(Click on the pictures to zoom in) Click here to reveal the answers. All lone pairs and hydrogens attached to carbon are shown. Drag diagonally to the opposite corner of the rectangle defined by the brackets and release the mouse, In this mode, up and down arrows will be displayed at various positions on the bracket. Formal charges may be associated with atoms in Lewis structures. Assign the formal charge to all atoms and determine the overall charge of the molecule. Add a structure to the canvas and then add brackets around it by pressing the mouse down where the first corner of the bracket will start. To place brackets a structure, use the bracket from the toolbar. To show the charge on an ion in ChemDoodle, use the bracket tool described below. In order to use the formula charge formula, we must observe the Lewis Dot Structure for the. In a Lewis structure, the charge on an ion is placed as a superscript on surrounding square brackets, as shown for the sulfate ion in the drawing to the right. Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. The overall ion charge is a property of an ion as a hole and is not associated with a particular atom. One can calculate the formal charges for any given atom with the help of the following formula: F. These charges help in knowing if the given structure of the molecule is stable or not. To show the formal charge on an atom in ChemDoodle, use the + or - tools that have been described previously. Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. For example, in the drawing of the sulfate ion (SO_4^2-) shown on the left, each oxygen atoms is assigned a formal charge of minus one and the sulfur atom is assigned a formal charge of plus two. Transcribed image text: Formal charges may be associated with atoms in Lewis structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
